Spectroscopy Wavelength Ranges: VIS, NIR, SWIR, MIR and FIR
Spectroscopy methods are defined primarily by the wavelength region of the electromagnetic spectrum used to probe materials. Different spectral regions interact with matter through different physical mechanisms, ranging from electronic transitions in the visible region to molecular vibrations in the infrared. These interactions determine which chemical bonds and material properties can be measured.
This article provides a technical overview of the wavelength regions used in spectroscopy, their physical basis, and the spectral ranges commonly used in industrial instruments and hyperspectral imaging systems.
Table of contents
Electromagnetic Spectrum Overview
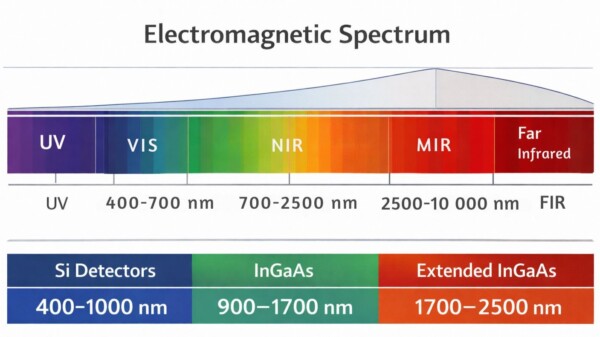
Spectroscopic techniques typically operate across the visible and infrared portions of the electromagnetic spectrum. These regions span wavelengths from roughly 400 nm to 1000 µm, with progressively lower photon energies at longer wavelengths.
| Region | Typical wavelength range | Primary interaction |
|---|---|---|
| Visible (VIS) | ~400–700 nm | Electronic transitions |
| Near-infrared (NIR) | ~700–2500 nm | Overtone / combination vibrations |
| Short-wave infrared (SWIR) | ~700–1700 nm | Often used subset of NIR |
| Mid-infrared (MIR) | ~2500–10 000 nm | Fundamental molecular vibrations |
| Far-infrared (FIR) | ~12 000–100 000 nm | Rotational and lattice vibrations |
The visible region transitions into the infrared at approximately 700 nm, where electronic absorption becomes less dominant and vibrational interactions begin to appear. (NLIR)
Infrared spectroscopy is therefore typically divided into NIR, MIR and FIR because these regions probe different molecular processes.
VIS vs NIR vs MIR Spectroscopy
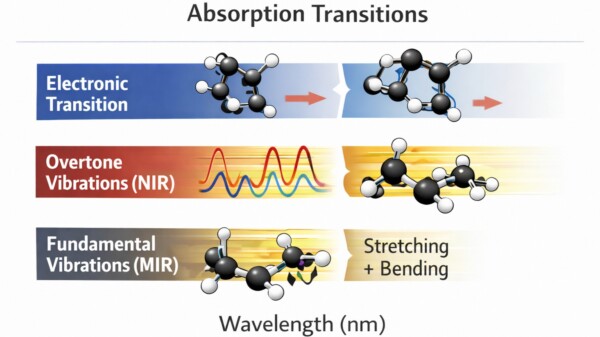
Although the visible and infrared regions are contiguous, the underlying physics of absorption is different.
| Spectral region | Wavelength range | Dominant transitions | Typical applications |
|---|---|---|---|
| VIS | ~400–700 nm | Electronic transitions | color analysis, pigments |
| NIR | ~700–2500 nm | Overtone & combination vibrations | composition, moisture |
| MIR | ~2500–10 000 nm | Fundamental vibrations | molecular identification |
Figure: Energy transitions measured in spectroscopy including electronic transitions (VIS), overtone vibrations (NIR), and fundamental vibrations (MIR).
Visible spectroscopy
In the visible region, photons excite electronic transitions in atoms and molecules. Absorption features correspond to chromophores such as transition metal complexes, dyes, or conjugated organic structures.
Industrial uses include:
- color measurement
- coating thickness
- pigment identification
- surface inspection
Near-infrared spectroscopy
NIR spectroscopy measures weak overtone and combination bands of molecular vibrations. These arise from bonds containing hydrogen.
Key absorbing bonds include:
| Bond type | Typical vibrational features |
|---|---|
| O–H | strong NIR overtone bands (moisture, hydroxyl groups) |
| C–H | hydrocarbons, polymers, oils |
| N–H | proteins, amines |
These bands originate from fundamental vibrations located in the MIR region but appear at shorter wavelengths as higher-order overtones. (Oxford Instruments)
Mid-infrared spectroscopy
MIR spectroscopy probes fundamental vibrational modes of molecules.
These are much stronger absorption features than NIR and provide detailed molecular identification, making MIR spectroscopy common in:
- FTIR chemical analysis
- gas sensing
- polymer identification
- pharmaceutical analysis
How NIR Spectroscopy Works
Near-infrared spectroscopy measures absorption of broadband light interacting with molecular vibrational overtones.
The typical measurement sequence is:
- A broadband light source illuminates the sample.
- Molecular bonds absorb specific wavelengths.
- Reflected or transmitted light is measured by a spectrometer.
- Multivariate models convert spectra into chemical or physical parameters.
Because NIR bands are weak and overlapping, spectral interpretation typically relies on chemometric modeling rather than single absorption peaks.
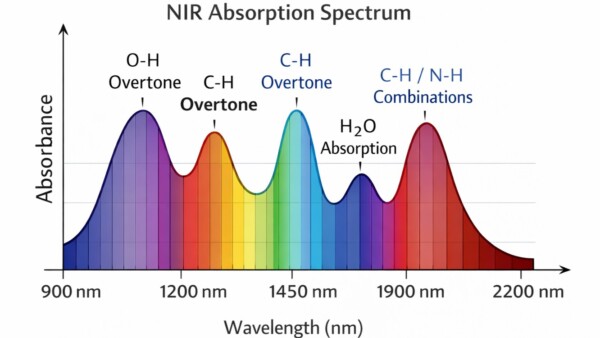
Key molecular absorption groups
NIR sensitivity arises primarily from hydrogen-containing bonds:
| Bond | Fundamental vibration (MIR) | NIR overtones |
|---|---|---|
| O–H stretch | ~2.7–3.2 µm | ~700–1500 nm overtones |
| C–H stretch | ~3.3–3.5 µm | ~1100–1800 nm |
| N–H stretch | ~2.8–3.1 µm | ~1000–1600 nm |
These absorptions allow NIR spectroscopy to quantify:
- moisture
- organic content
- hydrocarbons
- proteins
- polymers
Common Industrial Spectrometer Wavelength Ranges
Industrial NIR spectrometers typically operate in wavelength windows determined by detector technology.
Typical measurement ranges
| Spectrometer range | Detector type | Typical applications |
|---|---|---|
| 900–1700 nm | InGaAs | moisture, plastics, food |
| 1350–2150 nm | extended InGaAs | organic compounds |
| 1600–2400 nm | extended InGaAs | polymers, hydrocarbons |
| 900–2400 nm | dual detector systems | broad compositional analysis |
These wavelength ranges cover the most diagnostically useful overtone bands of O–H, C–H and N–H bonds.
Industrial applications
Common industrial use cases include:
Agriculture and food
- moisture measurement
- protein content
- fat and carbohydrate analysis
Polymer manufacturing
- resin composition
- additive concentration
- curing monitoring
Process industries
- chemical composition monitoring
- raw material verification
- blending control
Because NIR light penetrates deeper into materials than visible light, it is well suited for bulk composition measurements.
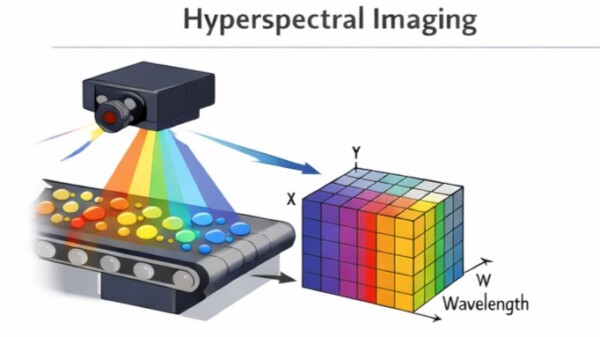
Hyperspectral Camera Wavelength Ranges
Hyperspectral imaging systems combine spectroscopy with imaging, producing a full spectrum for each pixel.
Typical wavelength ranges depend on detector technology.
Common hyperspectral spectral bands
| Camera type | Wavelength range | Detector | Applications |
|---|---|---|---|
| VIS–NIR | 400–1000 nm | silicon | color, agriculture |
| NIR | 700–2500 nm | InGaAs | material sorting |
| SWIR | 700–1700 nm | extended InGaAs | plastics, minerals |
| MIR | 2500–10 000 nm | MCT | chemical identification |
Silicon sensors typically cover 400–1000 nm, while InGaAs detectors extend sensitivity into the SWIR region up to roughly 1700–2500 nm. (iws.fraunhofer.de)
Typical hyperspectral applications
Food inspection
- foreign object detection
- bruising detection
- moisture mapping
Recycling and waste sorting
- polymer identification
- black plastic detection
- mixed material separation
Mining and geology
- mineral classification
- ore composition
Industrial inspection
- coating thickness
- contamination detection
Hyperspectral imaging is widely used where spatially resolved chemical information is required.
Limitations of NIR Spectroscopy
Despite its versatility, NIR spectroscopy has several technical limitations.
Weak absorption features
NIR absorption bands are overtones of MIR vibrations, making them much weaker than MIR features. This leads to:
- lower chemical specificity
- overlapping spectral peaks
Chemometric dependence
Because spectra contain overlapping bands, NIR measurements typically require:
- calibration models
- multivariate regression
- large training datasets
Without proper calibration, direct interpretation is difficult.
Limited molecular specificity
Unlike MIR spectroscopy, NIR does not provide unique molecular fingerprints. As a result:
- different compounds can produce similar spectra
- models may not transfer across materials
Sensitivity to physical effects
NIR spectra can also be affected by:
- particle size
- surface scattering
- temperature
- optical path length
These factors introduce variability that must be compensated through preprocessing and calibration.
Conclusion
Spectroscopic techniques operate across different wavelength regions of the electromagnetic spectrum, each probing different physical interactions with matter.
- VIS spectroscopy measures electronic transitions.
- NIR spectroscopy detects overtone vibrations of hydrogen-containing bonds.
- MIR spectroscopy measures fundamental molecular vibrations.
Industrial spectrometers commonly operate in ranges such as 900–1700 nm or 900–2400 nm, while hyperspectral cameras extend from visible wavelengths up to the mid-infrared.
Understanding these wavelength ranges and their molecular interactions is essential for selecting the appropriate spectroscopy technique for material analysis and process monitoring.
FAQ
FAQ1: What is the NIR wavelength range?
The near-infrared (NIR) wavelength range in spectroscopy is typically defined as approximately 700 to 2500 nm. This region is used to measure overtone and combination vibrations of molecular bonds, especially O-H, C-H, and N-H bonds.
FAQ2: What is the difference between NIR and SWIR?
SWIR is commonly treated as a subset of the broader NIR region. In the page content, SWIR is shown as approximately 700 to 1700 nm, while NIR extends to about 2500 nm.
FAQ3: What wavelengths are used in spectroscopy?
Spectroscopy uses different wavelength regions depending on the measurement principle. The page covers VIS (~400–700 nm), NIR (~700–2500 nm), SWIR (~700–1700 nm), MIR (~2500–10,000 nm), and FIR (~12,000–100,000 nm).
FAQ4: What does NIR spectroscopy measure?
NIR spectroscopy measures weak overtone and combination bands of molecular vibrations. It is especially sensitive to hydrogen-containing bonds such as O-H, C-H, and N-H.
FAQ5: What are typical NIR absorption bands?
Typical NIR absorption bands are associated with O-H, C-H, and N-H bonds. The page notes approximate overtone regions of 700–1500 nm for O-H, 1100–1800 nm for C-H, and 1000–1600 nm for N-H.
FAQ6: What are common industrial NIR spectrometer wavelength ranges?
Common industrial NIR spectrometer ranges listed on the page include 900–1700 nm, 1350–2150 nm, 1600–2400 nm, and 900–2400 nm, depending on detector design and application.
FAQ7: What is the difference between VIS, NIR, and MIR spectroscopy?
The page distinguishes the regions by their dominant physical interactions: VIS measures electronic transitions, NIR measures overtone and combination vibrations, and MIR measures fundamental molecular vibrations.
FAQ8: What are common NIR spectroscopy applications?
The page associates NIR spectroscopy with applications such as moisture measurement, composition analysis, protein and organic content analysis, polymer monitoring, and process control.